Hox Genes and the Evolution of Snake-Body Form
A Study Summary
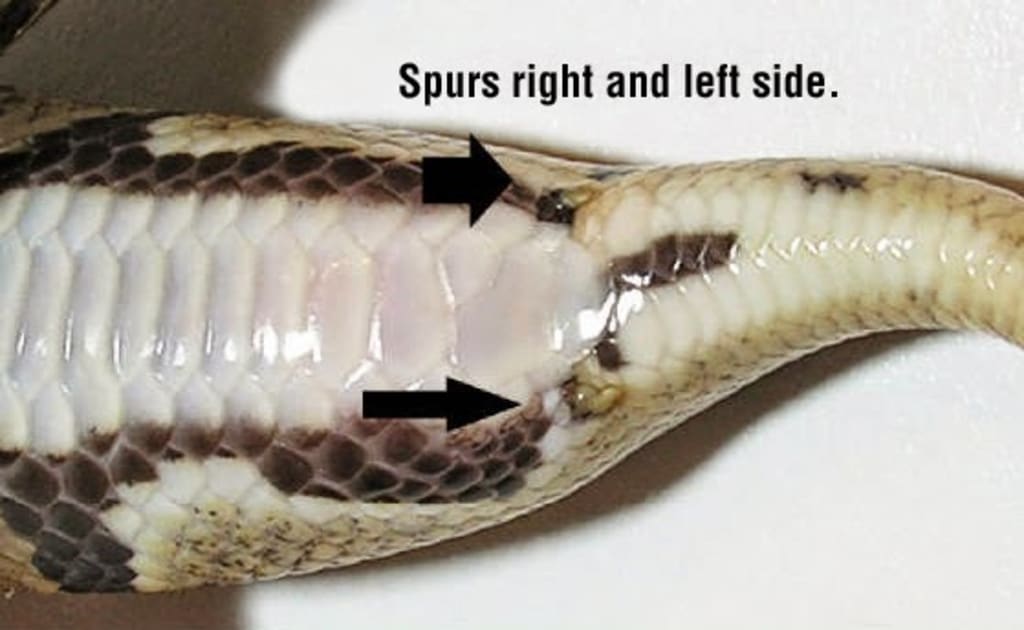
Abstract: Regulation of the vertebrate axial skeleton and the driving force behind the evolution of new body forms are due to Hox genes. Many hypotheses over the years have risen using Hox genes to explain snake-body form evolution. This paper will discuss some of those hypotheses.
Introduction
All throughout history, mythology, and religion, snakes have had a mostly bad image, but recently scientists have started considering the potential of adding snakes to the list of model organisms (Guerreiro and Duboule 2014). Snakes are of interest to the scientific community because of the controversy as to how their body form evolved, as well as what they evolved from. With multiple hypotheses floating around, it’s nearly impossible to determine whose hypothesis is correct. The evolution of snakes is difficult to research, but some scientists have tried different approaches. One approach is to decipher snake evolution by looking at samples of snake relatives, both from museums and living relatives such as lizards, other reptilians (Weins and Slingluff 2001), mice, and caecilians (which are classified as amphibians; Woltering et al. 2009).
There are different hypotheses about the ancestors of snakes. Some say that they likely came from ancient lizards that changed their morphology and physiology due to a burrowing lifestyle (Guerreiro and Duboule 2014). Diverse types of snakes fall somewhere on an evolutionary scale between primitive and advanced (Lee and Scanlon 2002). Pythons are one snake that supports this hypothesis. Pythons do not have legs, but they do show remnants of hind-limbs. These vestigial hind-limb are thought to have remained to help with breeding (Guerreiro and Duboule 2014). However, unlike the snake’s tetrapod relatives, the snake’s limb-buds are lacking an apical ectodermal ridge – the signaling center that usually controls the proximal to distal growth of limb buds (Guerreiro and Duboule 2014). Two approaches are taken when determining the best way to tackle the evolution of snake-body form: satisfaction from researching everything about one type of snake (potential model organism) and not being satisfied until every snake on the evolutionary scale has been fully researched. Both of those approaches have one thing in common: the role of Hox genes.
Hox genes are a focus of interest when determining the evolution of the snake form. During embryonic development, Hox genes are responsible for the patterning and morphogenesis of the vertebrate skeleton (Woltering et al. 2009; Gomez and Pourquie 2009). For example, the Hox10 gene controls somatic processing patterns that lead to rib development inhibition (Guerreiro et al. 2013). In most cases, Hox genes are nested, and anterior and posterior gene domains tend to overlap (Woltering et al. 2009). There is controversy in the scientific community on whether these Hox genes play a major or a minor role in the snake-form evolution.
Axial Skeleton Evolution Hypotheses
The axial skeleton is composed of the skull, hyoid bone, vertebral column, ribs, and sternum. It serves to protect head, neck, and trunk organs as well as support them. In the basis of domain mapping and transgenic expression, two hypotheses have been proposed to address the role of Hox genes in the evolution of the axial skeleton in snakes. The first hypothesis is that Hox expression is being modified upstream, causing a loss in regionalization, and a shift forward of HoxC6 and HoxC8 to the first post-atlanto-axial vertebral position is also either involved or is the result of the upstream modification (Head and Polly 2015). The second hypothesis states Hox expression shifts aren’t the cause of loss of regionalization, but rather it is caused by the downstream changes in cis-regulation (Head and Polly 2015; Woltering et al. 2009).
Neck-Trunk Body Regionalization Hypotheses
The neck-trunk regionalization of snakes is also an area of interest; for example, where does one region end and the other begin? There are a few hypotheses having to do with the neck-trunk body regionalization: The third hypothesis states that snake necks are really elongated (Tsuihiji et al. 2012). This hypothesis stems from the observation of snakes having typical anterior organs located more posteriorly (i.e. lungs and heart) which then led to the suggestion that snakes originate from aquatic environments (Tsuihiji et al. 2012). The fourth hypothesis states that snakes are completely lacking in the cervical region (Tsuihiji et al. 2012); as suggested by Cohn and Tickle (1999) who observe that the expression patterns of HoxC8 and HoxC6, typically located anteriorly, are found posteriorly in snakes such as pythons. The fifth hypothesis states that the various anatomical structures associated with neck-trunk boundary are displaced in snakes as compared to squamates with well-developed limbs (quadrupedal squamates). This hypothesis was developed when scientists noticed that multiple anatomical structures were displaced in snakes as compared to quadrupedal squamates (Tsuihiji et al. 2012).
Body Evolution from Lizards to Snakes Hypothesis
Another hypothesis arose from the curiosity regarding snake evolution from lizards in a phylogenetic analysis approach; The sixth hypothesis states that the elongation in snake-like squamates and the numerical increase in presacral vertebrae are related (Wiens and Slingluff 2001). A study done in 1999 by Cohn and Tickle suggest that elongate, limb-less species should also lack limb girdles due to their association with anterior expansion of midbody Hox genes (Wiens and Slingluff 2001). Gomez and Pourquie (2009) continue on to suggest that a shift in Hox gene expression along the AP axis would cause the absence of forelimbs and an increased number of thoracic vertebrae, as demonstrated in pythons.
Results
Woltering and colleagues (2009) used corn snakes and bearded dragons to analyze Hox gene expression in detail. They found that corn snakes lacked limb girdles, forelimbs, hindlimbs, sternal elements, and homogeneous ribs (except for the first three most anterior pre-cloacal vertebrae; Woltering et al. 2009). Woltering and colleagues (2009) found that the dorsal region of snakes extends for most the body, and even though there is poor regionalization they do detect collinear expression of Hox genes in the dorsal region, which have been linked to thoracic and lumbar regional patterning in mice and chickens. HoxC6, HoxA7, HoxC8, and HoxB9 (expressed more posteriorly) tend to show fading over approximately 10-20 somites without a defined anterior expression boundary (Woltering et al. 2009). Furthermore, Guerreiro and colleagues (2013) found that there is a single base pair change in the Hox/Paired box (Pax)-responsive enhancer in snakes that prevents Hox proteins from binding, which then prevents the blockage of rib formation and allows the thoracic region to extend into Hox10-expression areas of snake embryos. They conclude that polymorphism in the Pax/Hox binding site of the H1 enhancer, which is controlling the Myf5 gene, is directly related to the extended rib development in snakes, and they suggest it is a key factor in snake-form evolution (Guerreiro et al. 2013).
In other squamates, neck-trunk is marked by the forelimb girdle position. However, in corn snakes there isn’t a well-defined transition from neck to trunk. Wiens and Slingluff (2001) say that Cohn and Tickle (Hypothesis 6) were incorrect; the midbody Hox genes aren’t completely accountable for the phenomena. Their experiment shows that the process comes in steps: digit loss is correlated with limb reduction, and limb reduction is associated with body elongation. Woltering and colleagues (2009) suggests that the second hypothesis (above) is correct; Hox genes take on a different response due to altering the cis-regulatory sequence, thereby causing a downstream gene response. They also say that the differential activation of target genes could be dictated by the altering of Hox coding sequences (Woltering et al. 2009). Furthermore, Guerreiro and colleagues (2013) suggests that high expression levels of Hox6 throughout the entirety of the rib domain plays a key role in effective rib production in snake trunks.
Conclusion
There is still a great deal of research that needs to be done to determine the evolution of the snake-body form and the role of Hox genes. Out of the research conducted, most of the conclusions agree with a mix of all the hypotheses stated above. There isn’t a single correct hypothesis that can explain the exact role of Hox genes throughout the evolution of the snake-body form. Perhaps through studying various snake types (i.e. pythons, corn snakes) in depth, more can be determined about the topic, but relying on one snake type to find all the answers isn’t enough – there definitely needs to be variety to understand the role of Hox genes and to understand which genes may be specific towards a certain snake group as compared to what genes are common across all snake types, as well as other species that have a snake-form, (i.e. caecilians).
Table 1: This table shows the area of mutation (-) in the enhancer that controls limb development in pythons compared to other organisms in regions A, B, and C (Chambers 2016).
Region A - Organism Genome Sequence
Human TA-------AGCAACATCCTGACCAATTATCCAAACCATCCAGACA
Mouse TAA-TGACAGCAACATCCTGACCAATTATCCAAACCATCCAGCCA
Chinese Turtle TGAGCGACAGCAACATCCTGACCAATTATCCAAATTATCCAGACA
Alligator TGAGCGACAGCAACATCCTGACCAATTATCCAAATTATCCAGGCA
Anole TGTGCGACAGCAACATCCTGACCAATTATGCAAAGCATCCAGCA
Skink TATGCAACAGCAACATCCTGACCAATTATGCAAACTAGCCAGACA
Python C--------------------CCAATTGTGCAAAGCATCCAGACA
Region B - Organism Genome Sequence
Human ATT--TTAGGTAACTTCCTTTCTTAATTAATTGGACTGACCAGG
Mouse A--TCTGAGGTCACTTCCTCTCTTAATTAGTTGCACTGACCAGG
Chinese Turtle AATT-TGAGGTGACTTCCTTGCTTAATTAATTAATTAGATAGACCAGG
Alligator AATT-TGAGGTGACTTCCTAACTTAATTAATTAGGTAGACCAGG
Anole AATTCTGAGGTAACTTCCTTGCTTAATTAATTAGGTAGGCCAGG
Skink AATT-TGAGGTGACTTCCTTGCTTAATTAATTAGGTAGGCCAGG
Python AATT-T---------------TTAATTAATTAGGTAGGCCAGG
Region C - Organism Genome Sequence
Human CTGTGCTGGTGCTTGGAATGT--------CTATAAAGCTGAGCAA
Mouse CTTTGCTGG-GCTCAGGCTCT--------CCATAAAGCCAAGCAA
Chinese Turtle CAGTGCTGGTGCTCAGAATGT--------CTATAAAGCTGAGCAA
Alligator CAGTGCTGGTGCTCAGAATGT--------CTCTAAAGCTGAGCAA
Anole CTGTGTTGCTGCTAAGGATAT--------CTATAAAGCTGAGCAA
Skink CAGTGTTGGTGCTCAGAATAT--------CTATAAAGCTGAGCAA
Python CCAT-----------------CTATAAAGCTGTAAAGCTGAGCAA
About the Creator
Christina B
There's a story deep inside all of us






Comments
There are no comments for this story
Be the first to respond and start the conversation.